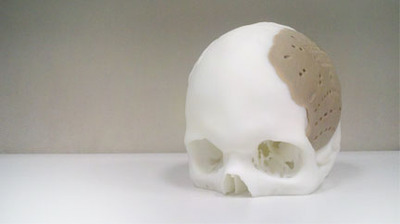
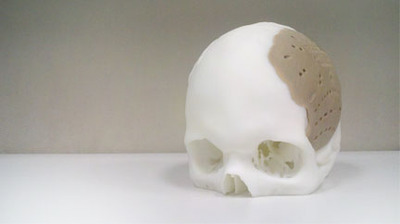
Oxford Performance Materials announced that it’s finally received US Food and Drug Administration clearance for their OsteoFab™ Patient Specific Cranial Device. This is a 3D printed medical implant designed to replace missing bone structures.
The advantage of this approach, is of course that the replacement parts can precisely match the missing elements. Extreme personalization, it is: the 3D printed part becomes part of you!
The parts are made from PEKK, a thermoplastic polymer.
Why is this approval notable? According to Oxford Performance Material’s press release:
FDA clearance of this device marks the first approval for an additively manufactured polymer implant.
We believe previous implants were exclusively metal, which while useful, come with side effects that are most notable at the security check X-Ray machine. Now your head can be made of plastic.